Colloidal particles exhibit equilibrium structures similar to atoms, such as crystals and liquids. The particles have sizes of around a micrometre, and suspended in a solvent, exhibit thermal energy kT like atoms do. We can furthermore be dressed with interactions similar to those of atoms, all leading to phases and overall behaviour similar to their atomic counterpart. Yet, as the particles are much larger, they can be easily observed by optical microscopy, providing fascinating insights into atomic structures and their formation dynamics. Besides being models, the particles serve as building blocks for micro- and nanostructured materials that have applications in medical science, photonics and optoelectronics.
Recent breakthroughs in particle fabrication allow synthesizing “patchy” particles that interact in specific directions, in a way similar to atomic orbitals. Indeed, we observe that these particles form structures analogous to molecules. The movie below (Fig. 1) shows a pair of particles with four patches (bright spots) in tetrahedral arrangement, clearly bonded via one of their patches. Remarkably, these 4-patch particles form structures similar to sp3-hybridized carbon atoms, including alkanes, such butane and butene, and cyclic compounds such as cyclopentane and cyclohexane, see Fig. 2. By observing these “colloidal molecules” directly in real space, we obtain unique insight into their atomic counterpart. Using confocal microscopy, we can even reconstruct the structures in three dimensions (see Fig. 3 for a reconstruction of the colloidal analogue of cyclopentane). We then analyse in detail their vibration spectrum, conformations and relaxation, and explore how this depends on the patch-patch attraction, allowing detailed insight into the statistical mechanics of molecules.
In this project, the student can explore the assembly of different molecular compounds, and study their formation and relaxation, thus obtaining insight into the molecular dynamics.

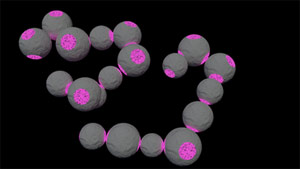
Fig. 1: Bonded tetrapatch particles.
The bonded particle pair exhibits Brownian motion, making it exhibit similar dynamics as thermally excited atomic molecules.
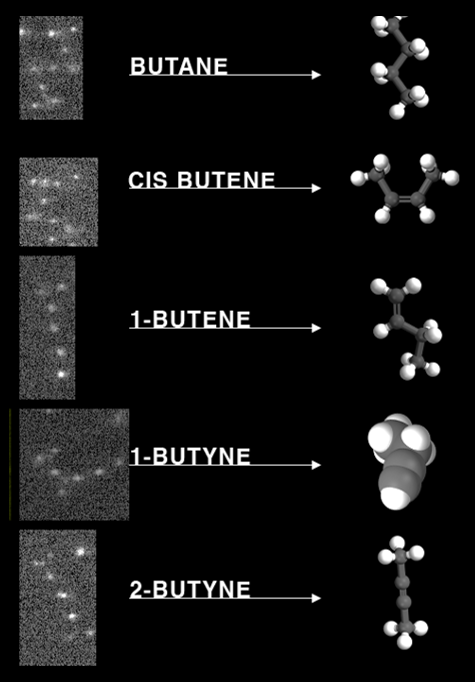
Fig. 2: Colloidal Molecules
Patchy particle structures observed by microscopy (left) and their molecular analogues (right). These 4-patch (tetramer) particles form molecular compounds known from carbon chemistry.
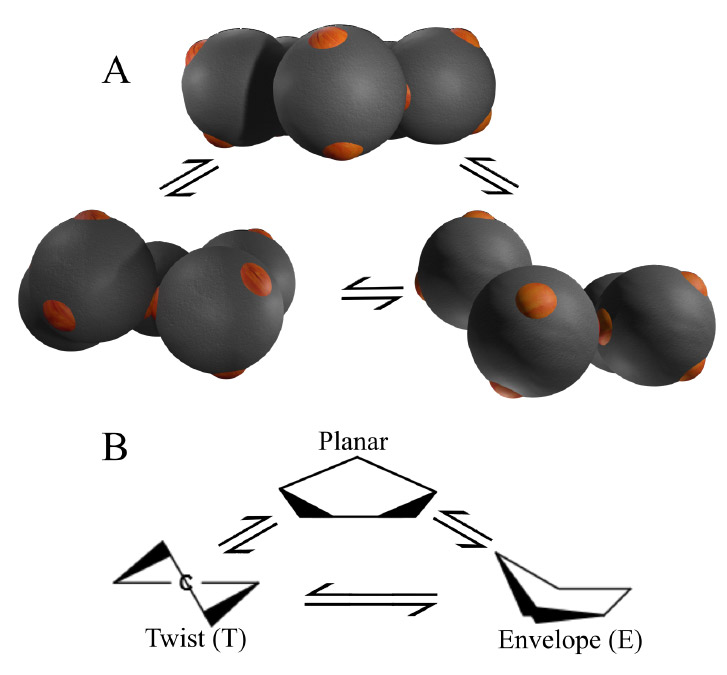
Fig. 3 Conformations of Colloidal Cyclopentane
Three-dimensional reconstruction of colloidal cyclopentane (A), and their molecular counterpart (B). The colloidal molecule shows planar, half-chair and envelope conformations, analogous to molecular cyclopentane. The larger (micrometer) size of colloidal cyclopentane, allows us to directly observe its dynamics in three dimensions.