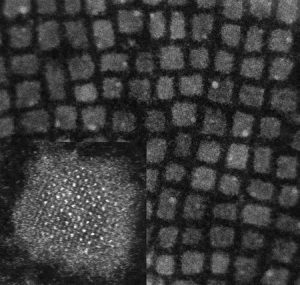
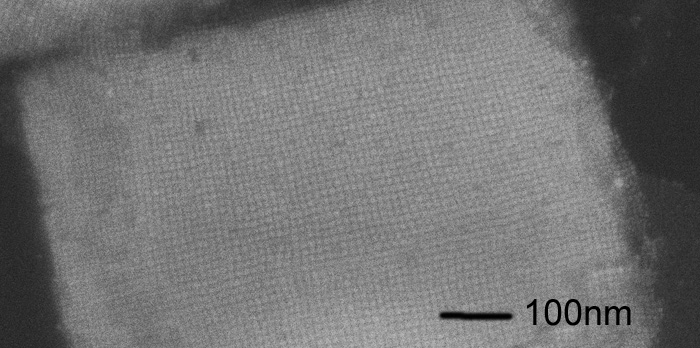
Nanocrystals offer fascinating properties and application perspectives for new photovoltaic devices: due to their small size, quantum effects become important, leading to discretized energy levels (-> electron in a box problem) just like atoms have. The advantage of nanocrystals is that the energy levels can be set by their size, and they can be assembled into “super” crystals, termed quantum-dot solids (see picture above). Just like atomic crystals, these supercrystals can exhibit a band structure if there is sufficient overlap of the quantum mechanical wave functions of neighbouring nanocrystals. The resulting “miniband” structure is analogous to the band structure of regular solids. Because the energy levels and overlap of the wave functions can be tuned, this offers fascinating opportunities to design band structures for applications in optoelectronic devices. The electronic states can be tailored by the strength of the nanocrystal interaction within the assembled quantum-dot solid, which is in principle determined by the QD distance. So far, however, this has remained a largely theoretical concept, and experimental realization of such tailored quantum-dot solids has remained a grand challenge.
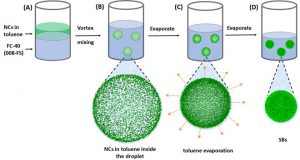
Here we take advantage of recently synthesized cubic all-inorganic perovskite QDs (Fig. 1), and the supercrystal assembly by emulsion templating (Fig. 2) to demonstrate the full transition from a single dot, via weakly and strongly coupled quantum solids to a fully joint solid, where due to fusion of the nanocrystals, the quantum confinement vanishes. The cubic perovskite NCs offer the great advantage that (i) their shape boosts self-assembly into highly-ordered cubic lattices enhancing mutual coupling, and (ii) they merge naturally by a diffusive mechanism if their stabilizing ligands get removed. The outcome of this fundamental research will be the bottom-up design of highly tunable new materials for efficient energy conversion.