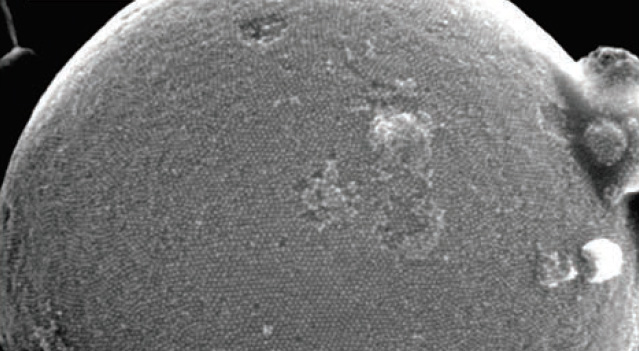
We investigate the assembly mechanism of quantum dots using x-ray scattering. This allows us to follow the nucleation and growth of quantum-dot crystals in situ (see cover image on the right)
Fig 1 below shows the x-ray images recorded during the crystallization of the quantum dots into superballs. The crystallization was achieved using emulsion templating: by suspending the q-dots in an oily solvent, and emulsifying the oil in water, we obtained small oil droplets that slowly evaporated through the water phase. By evaporating very slowly, over many hours, we finally obtain highly ordered crystals, as shown by the SEM image of the “superball” in Fig. 1e.
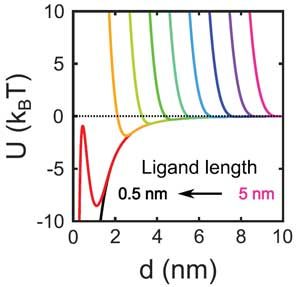
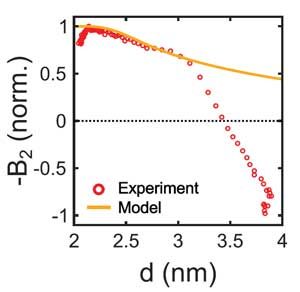
The time evolution of the structure factor is shown in Fig. 1g and h. These in-situ studies allow detailed insight into the crystallization process. For example, by evaluating in detail the scattering signal for small q, we obtain the compressibility and Virial coefficient of the quantum-dot solid. These studies reveal the evolution of the q-dot interactions during the assembly process. We can compare the determined virial coefficient with a pair interactions modelled based on Van der Waals attraction, and electrostatic repulsion between q-dots. Fig. 2.shows the interaction for decreasing ligand length during the evaporation (a), and a comparison of the resulting Virial coefficient with the experimentally measured one. Good agreement is observed for a range of distances.
Read our article in Advanced Materials

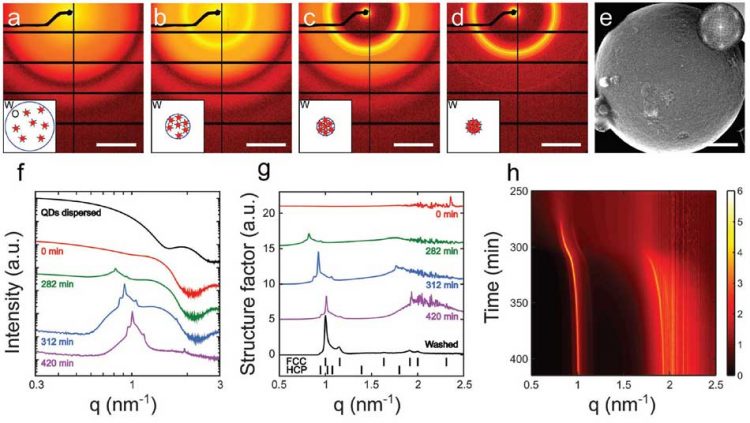
Figure 2. Quantum-dot interactions. Quantum-dot pair potential modelled from simple superposition of Van der Waals attraction and electrostatic repulsion for varying ligand length (a), and comparison of the resulting Virial coefficient with the experimentally measured one (b).