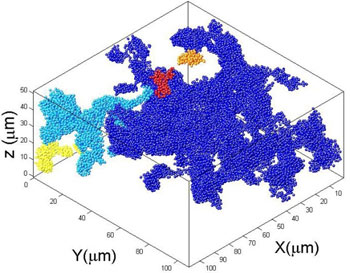
Glasses exhibit liquid structure, but solid properties. These materials are formed from the liquid by rapid quench, preventing the equilibrium crystal phase to form, and arresting the liquid phase, tremendously increasing its relaxation times until it appears as a solid. Besides glasses, there is a wide range of materials that are not in its equilibrium ground state, but rather in a kinetically arrested state, being metastable solids. Examples include gels, dense suspensions and emulsions, and networks of filaments or polymers. Many of these “soft materials” are used every day as foods and cosmetics, and constitute biological matter. Important questions are what makes these materials rigid, or flow to applied stress, and how can we understand aging, the slow evolution of their structure and properties?